Get A Free Legal Consultation
- We fight to maximize your results
- No out-of-pocket costs
- Over $11.7 billion recovered for families



The Allergan breast implant lawyers at Simmons Hanly Conroy are currently investigating cases involving the link between Allergan breast implants and BIA-ALCL.
If you or someone you love had breast implants removed due to a recall or have been diagnosed with BIA-ALCL, you may be able to file an Allergan breast implant lawsuit. If you have a case, you may be eligible for compensation.
In July 2019, Allergan, the pharmaceutical company that makes widely-used breast implants, issued a global recall of its BIOCELL textured breast implants and tissue expanders.
The recall came after the U.S. Food and Drug Administration (FDA) requested Allergan voluntarily pull the implants from the market due to a suspected connection to a certain type of cancer of the lymphatic system.
Since the recall, many patients who have been affected have come forward to file an Allergan breast implant lawsuit.
While the FDA has not recommended removing implants if no negative side effects are present, many patients are left scared about an increased risk of cancer and are wondering what to do.
Those who have chosen to file an Allergan breast implant lawsuit allege that implant removal comes at a significant expense, including pain and disfigurement. An Allergan breast implant lawsuit may help pay for costs associated with cancer or implant removal.
Contact us today to discuss a possible Allergan breast implant cancer lawsuit.


Breast implant cancer (breast implant-associated anaplastic large-cell lymphoma, or BIA-ALCL) is a type of non-Hodgkin’s lymphoma, which is a cancer of the immune system.
While it is considered rare, it has become more well-known in recent years.
BIA-ALCL is mainly found in the scar tissue and fluid near the breast implant. It can also spread throughout the body and can be deadly, especially if it is not promptly treated.
Symptoms of BIA-ALCL include:
Though the exact number of BIA-ALCL cases is not known, as of July 2019, the FDA received 573 reported cases. Of these cases, at least 33 patients have died.
If BIA-ALCL is discovered early, the prognosis is generally good. Surgery can usually be performed to treat the cancer, and chemotherapy or radiation therapy are sometimes recommended.
BIA-ALCL is not the same thing as breast cancer. BIA-ALCL is a cancer of the immune system that is usually slow-moving.
BIA-ALCL can usually be treated by removing both the breast implants and the scar tissue that forms around the implants. However, some patients have needed more extensive treatment.
Recent studies have shown that of the reported BIA-ALCL cases, roughly 80% have been linked to Allergan brand implants. Reports also show that Allergan’s BIOCELL products are six times more likely to cause cancer than other products.
Out of the 33 reported BIA-ALCL-related deaths, about 36% were associated with Allergan products.
Alarmingly, Allergan breast implant lawsuits have claimed that the manufacturer knew of the cancer risks long before the 2019 recall. The lawsuits allege Allergan began selling implants in 2006 but did not properly report breast implant patient safety concerns.
By the time better safety reporting requirements were put into place, thousands of breast implant patients had already experienced complications.
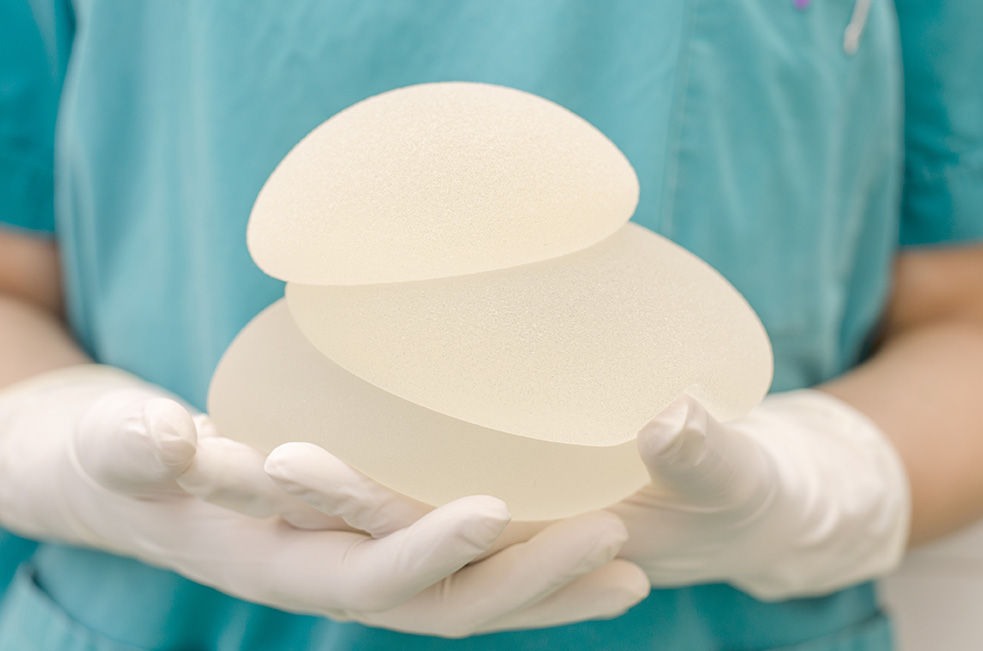
The FDA issued a Class 1 recall, which is considered the most serious type of recall since it is associated with serious injury or death.
In February 2020, Simmons Hanly Conroy Attorney and Partner Laura S. Fitzpatrick was appointed to the Plaintiffs’ Executive Committee (PEC) for the Allergan breast implant multidistrict litigation (MDL). This national committee of plaintiffs’ lawyers will handle breast implant cancer lawsuits filed by women around the country that are now consolidated in federal court in the District of New Jersey.
Laura has over a decade of experience helping victims from across the nation harmed by dangerous drugs and medical devices in mass tort litigation.
In 2019, she was part of the trial team that secured $325 million in settlements from several pharmaceutical companies to help counties in Ohio manage the ongoing opioid crisis. She also served on the trial team that litigated the bellwether trials in the Pinnacle defective hip implants MDL, which resulted in a $1 billion verdict.
As a member of the executive committee for the federal Allergan litigation, Laura is dedicated to helping victims of BIA-ALCL pursue compensation.

The Allergan breast implant lawyers at Simmons Hanly Conroy are currently investigating cases involving the link between Allergan breast implants and BIA-ALCL.
If you or someone you love had breast implant surgery and has since been diagnosed with BIA-ALCL, or the implants used have since been recalled, you may be able to file an Allergan breast implant lawsuit. If you have a case, you may be eligible for compensation.
To learn more about filing an Allergan breast implant lawsuit, contact Simmons Hanly Conroy.